
The theme of the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting is Advancing Equitable Cancer Care Through Innovation. From June 3 to 7 in Chicago, Illinois, and online, cancer researchers and clinicians from around the world will gather to discuss the latest research and how to ensure that all people receive the cancer care they need.
ON THIS PAGE
Some of the notable research that will be presented today includes:
Trastuzumab deruxtecan doubles progression-free survival in metastatic breast cancer with low HER2 expression levels
Who does this study affect? People with metastatic breast cancer that has low HER2 expression levels and has been treated with 1 or 2 types of chemotherapy.
What did this study find? Results from the DESTINY-Breast04 phase III clinical trial show that treatment with trastuzumab deruxtecan (Enhertu), when compared to standard-of-care chemotherapy, doubles progression-free survival among people with metastatic breast cancer that expresses low levels of HER2. The treatment also lengthens life for patients, regardless of hormone receptor status. This clinical trial also identified a new subset of metastatic breast cancer, called HER2-low, that can be effectively treated.
Metastatic breast cancer is breast cancer that has spread to distant parts of the body. There are different subtypes of breast cancer that are treated differently. One subtype of breast cancer is identified by whether the cancer expresses estrogen or progesterone receptors, called hormone receptor-positive (HR+) or hormone receptor-negative (HR–). Another subtype of breast cancer is determined by the expression of a protein called human epidermal growth factor receptor 2 or HER2. Typically, a breast cancer is considered HER2-positive if it expresses HER2 or HER2-negative if it does not. About 80% of metastatic breast cancer cases are considered HER2-negative, but researchers have found that 55% to 60% of those actually express low levels of HER2.
In this study, researchers wanted to know whether a targeted therapy treatment called trastuzumab deruxtecan could help patients with HER2-low breast cancer. Trastuzumab deruxtecan is a type of drug called an antibody drug conjugate or ADC. ADCs are drug combinations that work together to treat cancer. Trastuzumab targets the HER2 protein. Deruxtecan is a type of drug that targets a specific enzyme to kill cancer cells. This drug is already approved for the treatment of HER2-positive metastatic breast cancer after treatment with 2 or more HER2-targeted therapies.
This study included 557 people with HER2-low metastatic breast cancer. Of those, 373 were assigned to receive trastuzumab deruxtecan and 184 patients received standard-of-care chemotherapy. All study participants had HER2-low metastatic breast cancer and had received 1 or 2 lines of chemotherapy.
The study participants were followed for a median of 18.4 months. Among people with HR+ breast cancer and who received trastuzumab deruxtecan, the researchers saw a 49% reduction in the risk of the cancer growing and spreading and 36% reduction in the risk of dying, compared to the standard chemotherapy treatments.
Progression-free survival is the amount of time during and after treatment that the cancer does not grow or spread further. The progression-free survival doubled for people with HR+, HER2-low metastatic breast cancer who received trastuzumab deruxtecan compared to treatment with standard chemotherapy (10.1 months versus 5.4 months). For people with HR– breast cancer, trastuzumab deruxtecan also improved progression-free survival to 6.6 months, compared to 2.9 months for chemotherapy.
Participants who received trastuzumab deruxtecan also lived longer. The median overall survival for people in the HR+ group who received trastuzumab deruxtecan was 23.9 months, compared to 17.5 months for those who received chemotherapy. In the HR– group, the median overall survival for those who took trastuzumab deruxtecan was 16.6 months, compared to 10.3 months for chemotherapy.
There were fewer serious side effects among those who received trastuzumab deruxtecan, but some did develop serious lung problems.
What does this mean for patients? Metastatic breast cancer that expresses low levels of HER2 may also be effectively treated with trastuzumab deruxtecan, compared with standard chemotherapy.
"Our study shows that trastuzumab deruxtecan may be a new and highly effective targeted therapy option available for this newly defined patient population. It is important for patients to know what level of HER2 their cancer expresses, not just whether it’s positive or negative, especially as HER2-low status can be determined using commonly available tests.”
— lead study author Shanu Modi, MD
Memorial Sloan Kettering Cancer Center
New York, New York
Adding panitumumab to chemotherapy helps people with RAS wild-type metastatic colorectal cancer live longer
Who does this study affect? People with RAS wild-type metastatic colorectal cancer.
What did this study find? This phase III PARADIGM clinical trial found that combining the targeted therapy drug panitumumab (Vectibix) with standard chemotherapy as a first-line treatment helped people with metastatic RAS wild-type colorectal cancer live longer. Metastasis means that the cancer has spread to distant parts of the body. Wild-type means that there is not a mutation in the cancer’s RAS gene.
Colorectal cancer is the third most common cancer diagnosed in both men and women worldwide. When colorectal cancer has spread to distant parts of the body (metastasized), the 5-year-survival rate is 15%, and about 22% of patients are diagnosed at this stage. It has been 10 years since a new drug has shown an improvement over the current standard of care for metastatic colorectal cancer that does not have a RAS mutation.
This study compared the targeted therapy drugs panitumumab and bevacizumab (Avastin). Panitumumab targets the epidermal growth factor receptor (EGFR), and bevacizumab targets the vascular endothelial growth factor (VEGF) receptor. In addition to one of the targeted therapy drugs, every participant in this study received the current standard chemotherapy treatment, which is a combination of drugs called modified FOLFOX6 (mFOLFOX6). This drug combination includes folinic acid (leucovorin), fluorouracil (5-FU), and oxaliplatin (Eloxatin). The study included 823 people who had RAS wild-type metastatic colorectal cancer that had not been treated with chemotherapy. They were randomly divided into 2 groups: 400 participants received panitumumab plus mFOLFOX6 and 402 participants received bevacizumab plus mFOLFOX6. There were 21 patients who did not continue with the study.
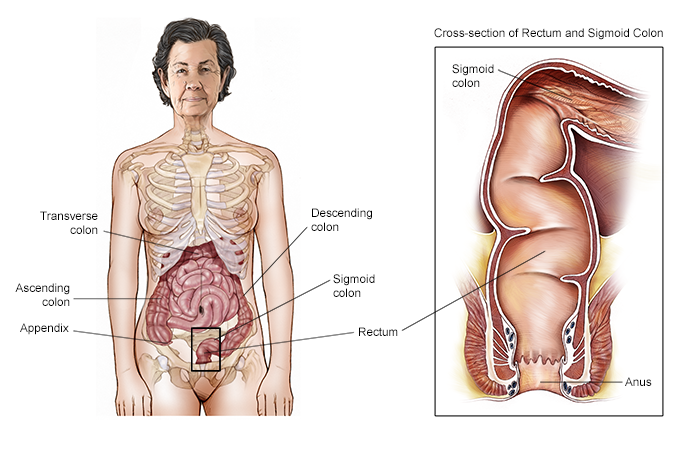
In addition to comparing the 2 drugs, the researchers also compared how well each treatment worked for people with a left-sided tumor compared to a right-sided tumor. Right-sided tumors develop in the cecum, the ascending colon, and in the transverse colon. Left-sided tumors develop at the curve where the transverse colon turns into the descending colon, called the splenic flexure because it is near the spleen. Left-sided tumors also develop in the descending colon and sigmoid colon. Where the tumor develops is important because previous research has found that these tumors have different characteristics and have different prognoses at later stages. Of the 400 patients who received panitumumab, 312 had left-sided tumors. Of the 402 patients who received bevacizumab, 292 had left-sided tumors.
Copyright 2004 American Society of Clinical Oncology. Robert Morreale/Visual Explanations, LLC.
To determine how well the treatments worked, researchers looked at several measures, including overall survival, progression-free survival, response rate.
The researchers’ main focus was on overall survival, which is how long people diagnosed with the disease are alive after treatment. Panitumumab improved overall survival for all study participants, and people with left-sided tumors had an 18% lower risk of death if they received panitumumab.
For people with left-sided tumors, the median overall survival rates were:
-
37.9 months for people treated with panitumumab
-
34.3 months for people treated with bevacizumab
Among all study participants, the median overall survival rates were:
-
36.2 months for people treated with panitumumab
-
31.3 months for people treated with bevacizumab
Researchers also looked at progression-free survival, which is the amount of time during and after treatment that the cancer does not grow or spread further. Both groups had similar rates of progression-free survival.
The percentage of people who had their tumor shrink or stop growing after treatment is called the response rate. Among those with left-sided tumors, panitumumab had an 80.2% response rate and bevacizumab had a 68.6% response rate. Among all participants, panitumumab had a 74.9% response rate and bevacizumab had a 67.3% response rate.
What does this mean for patients? The standard of care for RAS wild-type colorectal cancer has not changed in 10 years. Panitumumab plus mFOLFOX6, especially for left-sided tumors, appears to be an effective first-line treatment for people with RAS wild-type metastatic colorectal cancer. This study also shows the importance of genetic testing and tumor location for determining treatment options for these patients.
"This trial demonstrates that if gene testing shows that a tumor is a RAS wild-type, the choice of initial treatment with panitumumab with mFOLFOX6 chemotherapy is superior to initial treatment with bevacizumab with mFOLFOX6 chemotherapy for those people with left-sided tumors. It has long been believed that the sequence of metastatic colorectal cancer treatment does not matter as long as patients had access to the drugs at some point, which has now been disproven.”
— lead study author Takayuki Yoshino, PhD, MD
National Cancer Center Hospital East
Kashiwa, Japan
High-dose ifosfamide helps people with recurrent or refractory Ewing sarcoma live longer
Who does this study affect? People with Ewing sarcoma that comes back after treatment (recurrent) or that was not stopped by initial treatments (refractory).
What did this study find? The phase II/III rEECur clinical trial found that high-dose ifosfamide (Ifex) is a more effective chemotherapy treatment for people with recurrent or primary refractory Ewing sarcoma when compared to 3 other commonly prescribed chemotherapy treatments.
Ewing sarcoma is rare. About 200 children and teens are diagnosed with the disease each year in the United States. Ewing sarcoma tumors account for 1% of all cancers in children and teens younger than 15 and 2% of all cancers in teens between the ages of 15 and 19. For people with recurrent or primary refractory Ewing sarcoma, the 5-year survival rate is about 15%.
This is the first randomized trial to compare chemotherapy treatments for people with this diagnosis. The study participants ranged from ages 4 to 49, and the median age was 19 years old. The first phase of this study included 451 patients who were randomly chosen to receive 1 of 4 common chemotherapy regimens: topotecan (Hycamtin) plus cyclophosphamide (available as a generic drug), irinotecan (Camptosar) plus temolozomide (Temodar), gemcitabine (Gemzar) plus docetaxel (Taxotere), or high-dose ifosfamide. After early assessments, the combinations of irinotecan plus temolozomide and gemcitabine plus docetaxel were found to be less effective at treating the Ewing sarcoma and were removed from the study.
The next phase of the clinical trial only compared the high-dose ifosfamide and the combination of topotecan plus cyclophosphamide. There were 73 patients who received ifosfamide and 73 patients who received topotecan plus cyclophosphamide.
After a median follow-up of 40 months, researchers analyzed event-free survival, which is the amount of time after treatment ends that the cancer does not come back or get worse. For high-dose ifosfamide, event-free survival was 5.7 months. For the combination of topotecan and cyclophosphamide, it was 3.7 months.
The researchers also analyzed overall survival for both groups. For high-dose ifosfamide, overall survival was 16.8 months. For the combination of topotecan and cyclophosphamide, it was 10.4 months.
The most common serious side effects of both treatments included neutropenia, infections, vomiting, nausea, diarrhea, problems with brain function or structure, and kidney problems. When researchers looked at quality of life, they found that young patients who received ifosfamide had better quality of life than those who received the combination of topotecan and cyclophosphamide. For adults, no difference in quality of life was seen between the treatments.
What does this mean for patients? For people with Ewing sarcoma that has come back or that was not stopped by initial treatments, high-dose ifosfamide lengthens life when compared to other current chemotherapy regimens.
"The rEECur study has, for the first time, accrued randomized data for 4 widely used chemotherapy regimens and is now accruing data for a fifth regimen. Before the rEECur study, the basis for choosing drugs for patients with relapsed or refractory Ewing sarcoma was weak and lacking randomized trials to inform clinicians or patients about which treatments were most effective and/or most toxic.”
— lead study author Martin McCabe, MD, PhD
University of Manchester
Manchester, England
Stem cell transplant improves progression-free survival for younger people with newly diagnosed multiple myeloma
Who does this study affect? People who have been recently diagnosed with multiple myeloma.
What did this study find? The phase III DETERMINATION clinical trial found that including an autologous bone marrow/stem cell transplant early in the treatment plan for younger people with newly diagnosed multiple myeloma significantly improved progression-free survival. Progression-free survival is the amount of time during and after treatment that the cancer does not grow or spread further.
Multiple myeloma is a blood cancer that affects the plasma cells in the bone marrow. Bone marrow is the spongy tissue inside bones that creates different types of blood cells, including plasma cells. Plasma cells produce antibodies to help the body fight infection and they are an important part of the immune system. Multiple myeloma is most commonly diagnosed in people older than 60 and the median age at diagnosis is 70 years old.
The 722 participants in this study had newly diagnosed multiple myeloma and ranged in age from 18 to 65 years. They were randomly divided into 2 treatment arms, or groups.
-
Non-transplant group: This group received 3 cycles of RVd, treatment to move stem cells from the bone marrow to the bloodstream (called stem cell mobilization), and then 5 more cycles of RVd. RVd is a combination of 3 drugs used to treat multiple myeloma: lenalidomide (Revlimid), bortezomib (Velcade), and dexamethasone (multiple brand names). Lenalidomide is an immunomodulatory drug. Bortezomib is a targeted therapy. Dexamethasone is a steroid. After these cycles were complete, the people in this group received maintenance therapy with lenalidomide. The median age for the 357 people in this group was 57 years.
-
Transplant group: This group received chemotherapy with melphalan (Evomela), which is used to prepare the body for a stem cell transplant, and then the autologous stem cell transplant. After the transplant, they received 2 more cycles of RVd and maintenance therapy with lenalidomide. The median age for the 365 people in this group was 55 years.
Stem cell mobilization is done with medications so that stem cells can be collected from the blood and given back to the patient if they need a stem cell transplant in the future. For an autologous stem cell transplant, a person’s own stem cells are collected and then treatment with high-dose, intensive chemotherapy is given. This chemotherapy can damage a person’s stem cells and immune system. After chemotherapy ends, the person’s stem cells are reintroduced to the body to restore the body’s immune system and its ability to make blood cells and fight infection. The side effects of a stem cell transplant can be serious and challenging for patients, so some people choose to delay or not have a transplant.
After a median follow-up of 76 months, the myeloma had stopped progressing for people in the transplant group for a median of 67.6 months, compared with a median of 46.2 months for those in the non-transplant group. In the transplant group, 53% of the participants went on to need further treatment, compared with 63% of the participants in the non-transplant group. Among those who needed more treatment in the non-transplant group, 28% went on to have a stem cell transplant. There were 88 deaths in the transplant group and 90 deaths in the non-transplant group.
Serious, treatment-related side effects were more common among those who received the stem cell transplant. In the transplant group, 94% of participants had serious side effects, compared with 78% of participants in the non-transplant group. People in the transplant group reported lower quality of life scores than those in the non-transplant group, but their scores improved after the stem cell transplant.
What does this mean for patients? For younger people with multiple myeloma, initial treatment with an autologous stem cell transplant led to a median 21.4-month improvement in progression-free survival when compared with treatment without a stem cell transplant.
"The findings from DETERMINATION will have importance for real-world practice and the treatment of younger patients with multiple myeloma by informing providers and patients alike of the important considerations to be made in selecting treatment options in the newly diagnosed, transplant-eligible setting, as well as important implications for future research.”
— lead study author Paul G. Richardson, MD
Dana-Farber Cancer Institute
Boston, Massachusetts
Stay Informed
Visit the Cancer.Net Blog each day of the meeting for more scientific highlights from the 2022 ASCO Annual Meeting and how the new research will affect patient care. You can also keep up with the meeting’s news by following Cancer.Net on Facebook and Twitter, where you can follow the #ASCO22 hashtag.
Like what you’ve read here at Cancer.Net? Sign up for our monthly Inside Cancer.Net e-newsletter.
